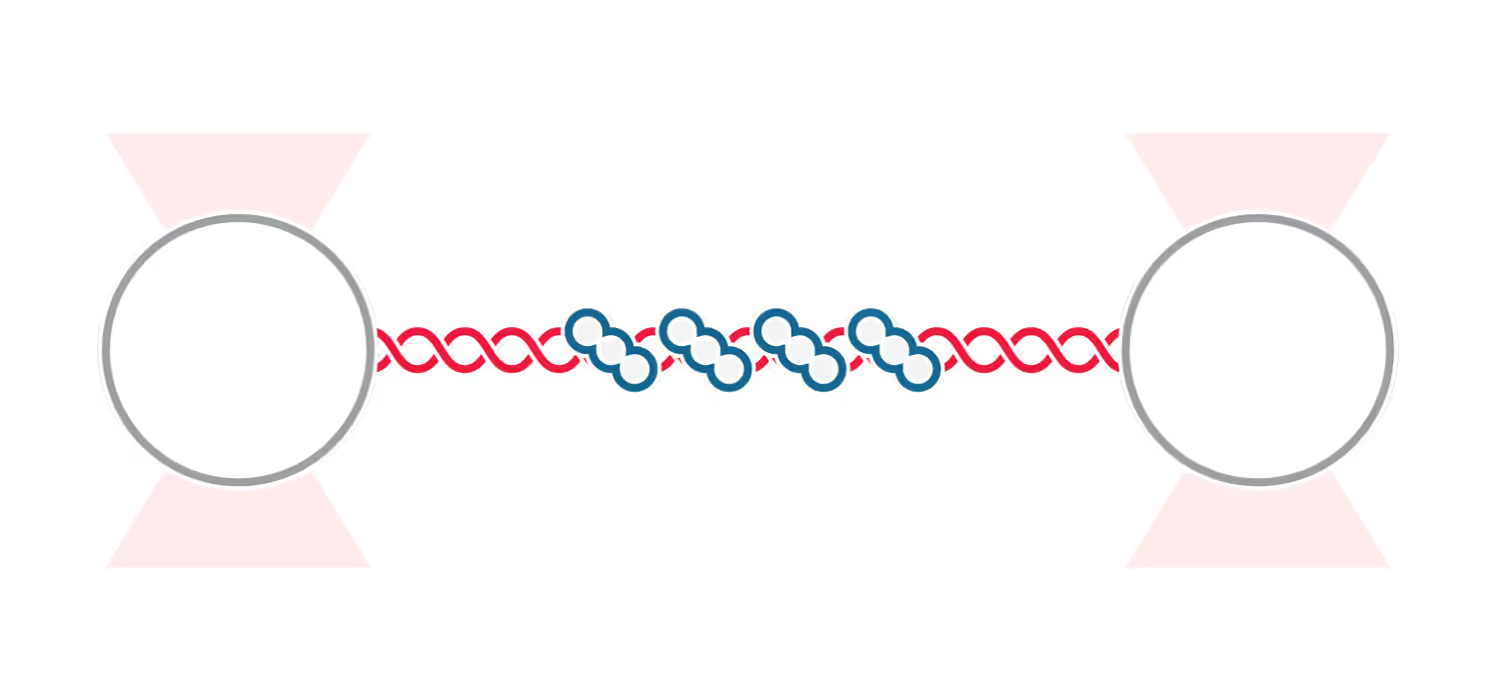
A recent publication in FEBS Letters describes how the presence of ADP and ATP affects the structural transitions that single-stranded DNA (ssDNA) undergoes upon RecA-filament assembly. The researchers used the u-Flux™ Laminar Flow Microfluidics combined with optical tweezers to demonstrate that the bacterial homologous recombination-enzyme exists in two interconvertible states.
The findings explain the dynamic properties of bacterial RecA-filament assembly and serve as a starting point to understand the properties of eukaryotic homologous recombination.
Alekseev et al. could readily measure the effect of active (presence of ATP) and inactive (presence of ADP) form of RecA filaments on an ssDNA by transitioning the optically trapped DNA between laminar flow channels. They found that exposing an active RecA–ssDNA filament to an ATP-free channel (with or without ADP) shortened the end-to-end distance of the ssDNA. These compressed states appeared to be reversible, as demonstrated by moving the tethered DNA molecule between the flow channels.
The researchers also measured the stability of RecA filaments upon exposure to the different nucleotide cofactors by measuring the ssDNA’s stiffness and found that RecA readily dissociated in the presence of ADP compared with ATP or no nucleotide cofactors.
Their findings provide new insights into how ATP hydrolysis induces structural and mechanical transitions associated with RecA filaments. According to the authors, demonstrating the role of interconvertible ADP- and ATP-dependent filament states on mechanical properties can help our understanding of recombination processes.
Congratulations to all the authors involved in this study for this exciting publication!
For more information, read the full article published in the journal FEBS Letters titled “Single-molecule analysis reveals two distinct states of the compressed RecA filament on single-stranded DNA“.