In a first-of-its-kind study from Leiden University, Evers et al. have provided insights into the mechanical properties of monocytes – cells crucial in the fight against infection.
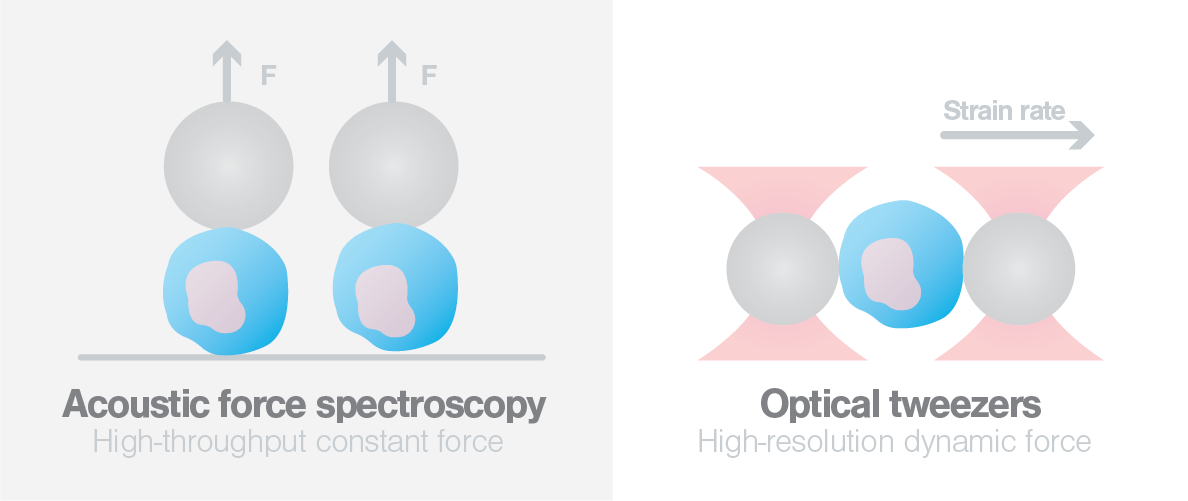
By combining two of LUMICKS’ single-cell force spectroscopy approaches, C-Trap® optical tweezers and acoustic force spectroscopy (AFS), they were able to determine that the mechanical properties of monocytes are affected by changes in temperature. Furthermore, exposing human monocytes to CCL2 – a chemokine and primary mediator in monocyte recruitment – can alter the mechanical properties of monocytes helping them switch to a migration-competent state and potentially progress to sites of infection.
Monocytes make up an important part of our innate immune system, as they reside in our blood and tissues and serve to protect our body against foreign infection. To move to a site of infection and exert an effective immune response, monocytes need to continuously adapt their shape so that they can pass through the narrowest capillaries and bifurcations in the circulation. Even though these functions depend on the mechanical properties of the cell, the mechanical behavior of monocytes is still poorly understood. This is what the scientists in this study set out to uncover.
As a first step, the researchers employed LUMICKS’ C-Trap optical tweezers to trap and stretch freshly isolated monocytes from healthy human blood bank donors. By performing multiple stretching cycles, they observed a strain-rate dependent increase in cell stiffness. They also observed that at room temperature (25°C) the monocytes showed a more solid-like behavior, whereas increasing the temperature to physiological conditions (37°C) reduced cell stiffness. These results, notably, show that temperature significantly impacts the mechanical properties of human monocytes, and underline the necessity to conduct cell mechanical studies under physiological conditions.
To delve deeper into the mechanical properties of the monocytes, the scientists then exposed the cells to the chemokine CCL2 (also known as monocyte chemoattractant protein 1). CCL2 is a chemokine well known for its role in monocyte recruitment to sites of inflammation, however, not much is known about its effects on the mechanical properties of cells. By exposing the cells to CCL2 and using the C-Trap to apply different strain rates, the researchers observed an increased cell stiffness, which was true only at higher strain rates. Strikingly, the increased stiffness was comparable to that observed with cells under physiological conditions (37°C). Moreover, with the help of AFS the authors showed that monocytes also vary in their mechanical properties just as they vary in their morphological heterogeneity (i.e., size, shape, and texture), and that this variability is regulated by CCL2. Taken together, these results showed for the first time that CCL2 can alter the mechanical properties of monocytes, which is crucial in their migration to sites of infection.
The results presented in this study – the first of its kind on monocytes – could give us fundamentally important insights into how monocytes alter their mechanical properties to switch from a resting state into a migration-competent state to fight human disease. Ultimately, this knowledge could provide a novel biomarker for health and disease.
To find out more about this study and learn how LUMICKS’ C-Trap optical tweezers can be applied in cell mechanics studies, read the full article “Single-cell analysis reveals chemokine-mediated differential regulation of monocyte mechanics”, published in iScience.