A recent publication in Science describes a new glass-like structural behavior that protein droplets can adopt, regulating their biochemical behavior as they age. Specifically, through rheological measurements using the C-Trap® optical tweezers, the researchers discovered material properties that may help cells respond to a dynamic molecular environment.
The findings offer essential insights into the time-dependent physical and functional properties of them, which can help researchers understand the physiology and pathological properties deriving from these changes.
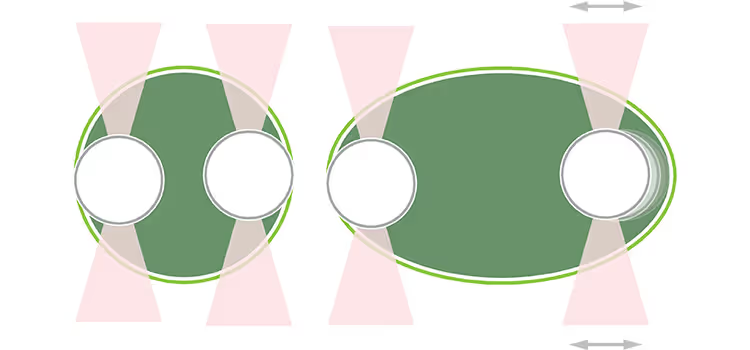
To study the time-dependent material behaviors of protein droplets, Jawerth et al. performed both active and passive rheology, which is the study of deformation and flow of a matter. They used optical tweezers for the active rheology assessments, linking two optically trapped beads to a droplet. The investigators next excited one of the beads with specific frequencies and measured the displacement of that bead to evaluate droplet deformation at different time points.
The team found that, at early time points, protein droplets demonstrated both viscous and elastic properties. By contrast, at later time points, the condensates lost their viscosity and became predominantly elastic. Further assessment with passive rheology, where researchers follow the movement of a fluorescent bead inside droplets, confirmed these findings.
These observations led the authors to describe the droplet behavior as a so-called Maxwell glass. The definition is based on the observed viscoelastic properties of the condensates, resembling a Maxwell fluid (with both elastic and viscous properties), but with age-dependent material properties seen among glass-forming structures.
The observed aging properties were caused by the reduced size of the droplets and subsequent increase in density, resulting in enhanced molecular interactions and stiffness. The authors suggest that this increased stiffness could impede the cellular environment.
“Cells in tissues are constantly responding to mechanical perturbation from other cells in the tissue and must be able to respond as soft materials,” the authors stated. “Glass-like aging of protein condensates may, therefore, offer cells a way to flexibly modulate mechanical properties of membrane-less organelles while allowing for rapid response to changing environmental cues.”
The findings highlighted in this study emphasize the added value of dynamic single-molecule to characterize structural properties properly.
According to the team, the “role of glass-like behaviors [has] not been well characterized in biology, which has tended to stress gel-like characteristics of biological materials.”
Here, they elegantly demonstrate how timepoint measurements with the C-Trap capture the dynamic behaviors of glass-like structures.
You can read the full article titled “Protein condensates as aging Maxwell fluids” in Science.